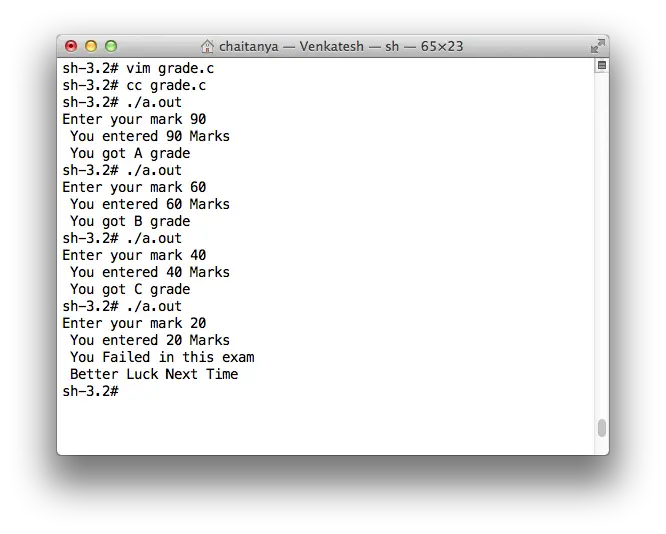
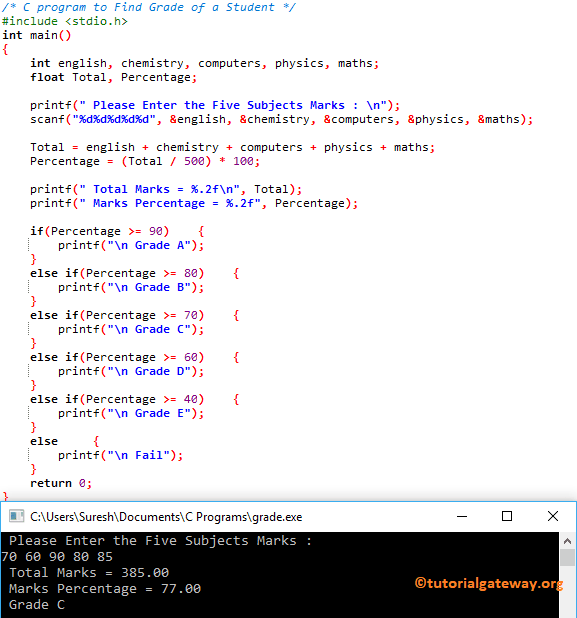
The ratio of mass percent of C and H of an organic compound (CxHYOZ) is 6:1 . If one molecule of the given compound (CXHYOZ) contains half as much oxygen as required

The ratio of mass percent of C and H of an organic compound (CXHYOZ) is 6:1 . If one molecule of the above compound (CXHYOZ) contain half as much oxygen as required