Calculate the mass of ascorbic acid (Vitamin C, C6H8O6) to be dissolved in 75g of acetic acid to.... - YouTube

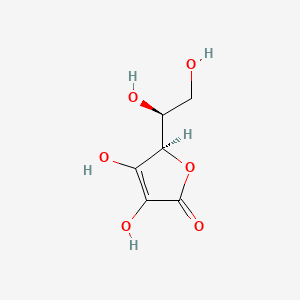
Calculate the mass of ascorbic acid (Vitamin C. C6H8O6 ) to be dissolved in 75 g of acetic acid to lower its melting point by 1.5^∘C. Kf = 3.9K kg mol^- 1 .

QUANTITIES, MOLES & EQUATIONS CONVICTIONS ARE MORE DANGEROUS THAN LIES. NIETZSCHE Ch 4.1 J.C. Rowe Windsor University School of Medicine. - ppt download

NCERT Solution Intext 2.11: Calculate the mass of ascorbic acid (Vitamin C, C6H8O6) to be dissolve - YouTube

Chapter Three: Stoichiometry: Calculations with Chemical Formulas and Equations Chapter Three: Stoichiometry: Calculations with Chemical Formulas and Equations. - ppt download

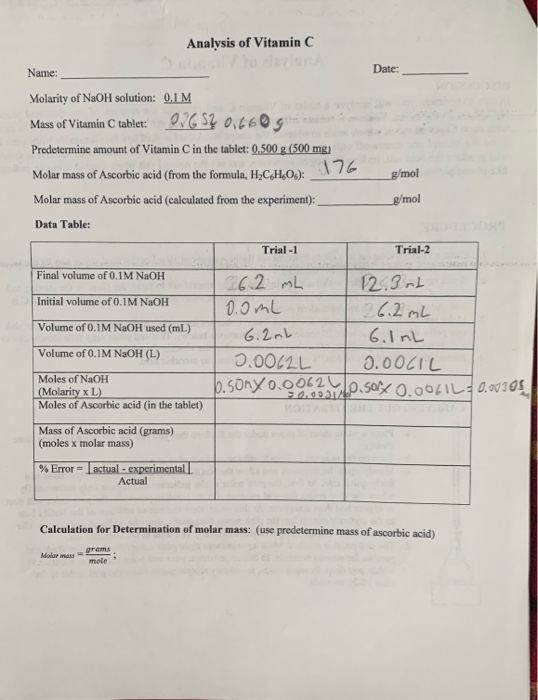
SOLVED: How many molecules of Vitamin C are in 500. mg of Vitamin € ( Molar mass of vitamin € = 176 g/mol)? 6.022*1023 1.71 X1023 1.71 x10 24 1.71 x1021
![Solved] Calculate the mass of ascorbic acid to be dissolved in 75g of acetic acid to lower its - Brainly.in Solved] Calculate the mass of ascorbic acid to be dissolved in 75g of acetic acid to lower its - Brainly.in](https://hi-static.z-dn.net/files/dcf/9c7caa88d0eb3f9e2f4686f7fccfbdb2.jpg)
Solved] Calculate the mass of ascorbic acid to be dissolved in 75g of acetic acid to lower its - Brainly.in

SOLVED: Ascorbic acid, vitamin C, is a water-soluble vitamin. Its chemical formula is C6H8O6 and its molar mass is 176.07 g / mol. A solution containing 80.5 g of vitamin C dissolved

SOLVED: The molecular formula of ascorbic acid (vitamin C) is C6H8O6. Determine the percent composition by mass in vitamin C (the percent of each constituent element).
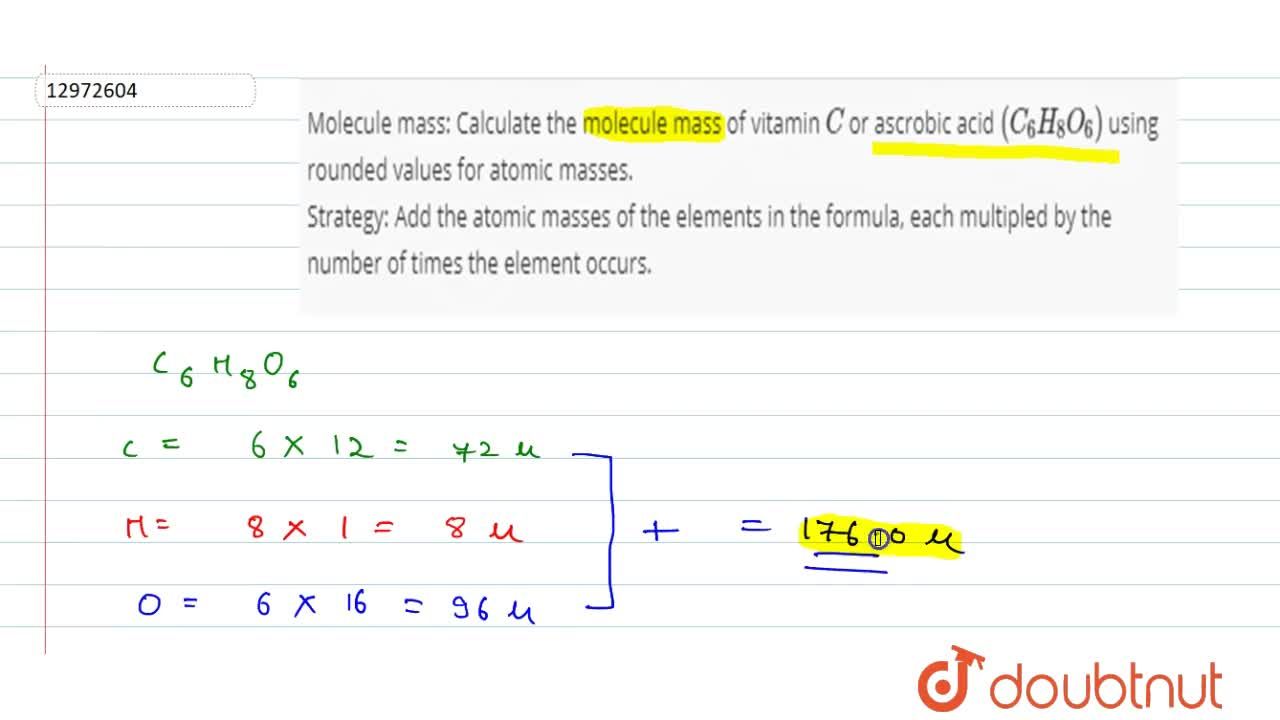
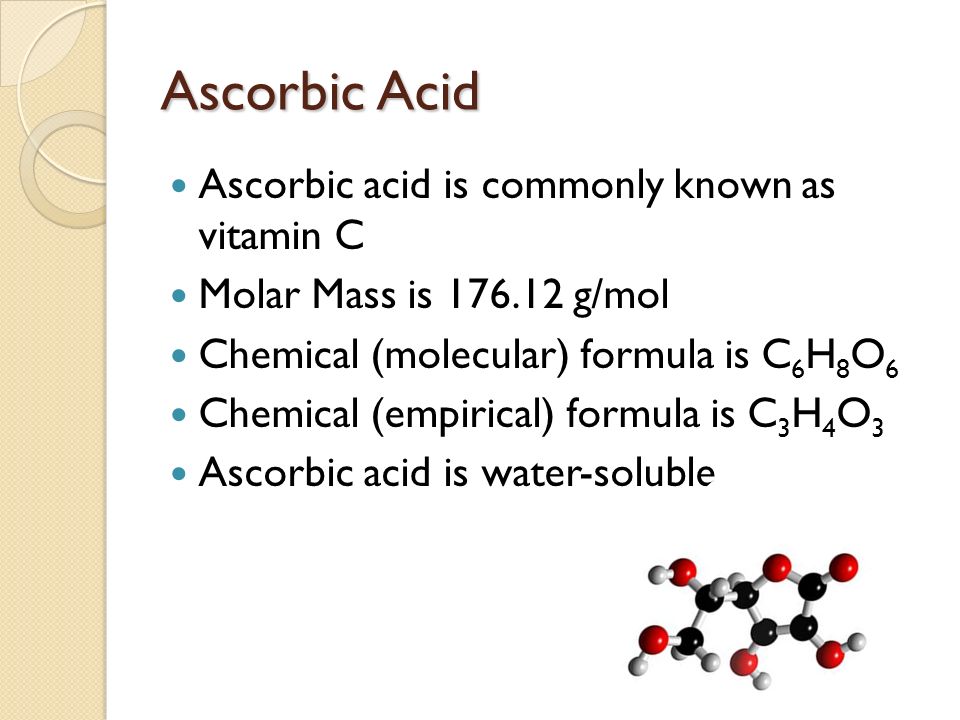
Molecule mass: Calculate the molecule mass of vitamin C or ascrobic acid (C(6) H(8) O(6)) using rounded values for atomic masses. Strategy: Add the atomic masses of the elements in the formula,
Calculate the mass of ascorbic acid (Vitamin C, C6H8O6) to be dissolved - Sarthaks eConnect | Largest Online Education Community

Calculate the mass of ascorbic acid (Vitamin C, C6H8O6) to be dissolved in 75 g of acetic acid to lower its melting point by 1.5^@C. Kf = 3.9 K kg "mol"^(-1) .

Calculate the mass of ascorbic acid (C6H8O6) to be dissolved in 75 g of acetic acid to lower its melting point by 1.5^0C . (Kf for acetic acid is 3.9 K kg mol^-1 ).

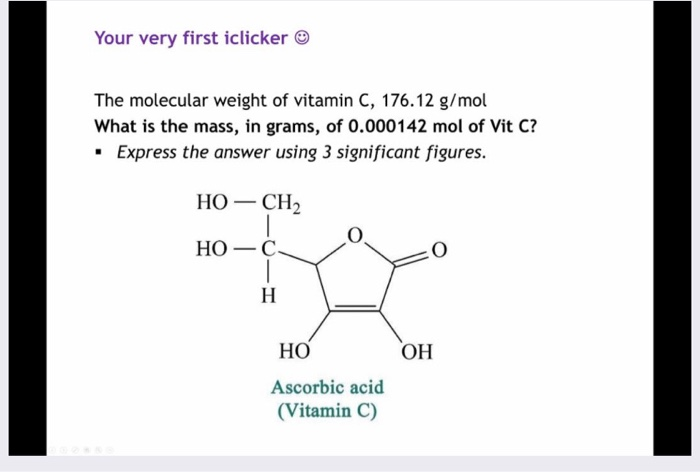
SOLVED: 'What is the mass, in grams, of 0.000142 mol of vitamin C? The molecular weight of vitamin C, C6H8O6, is g 176.12 mol What is the mass, in grams, of 0.000142