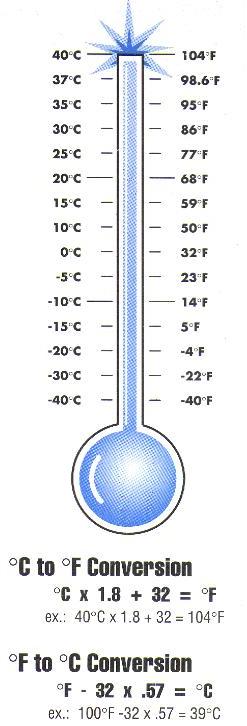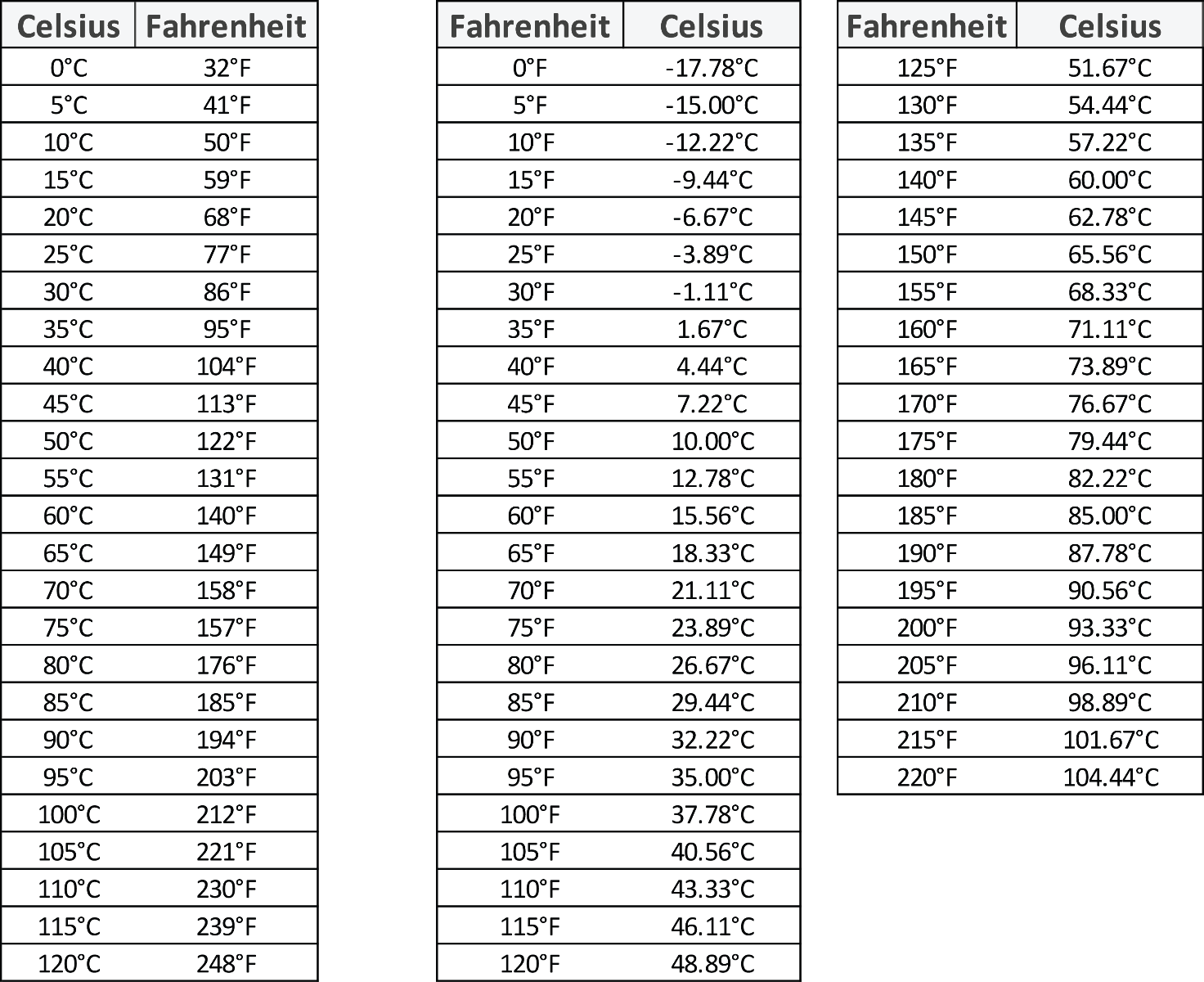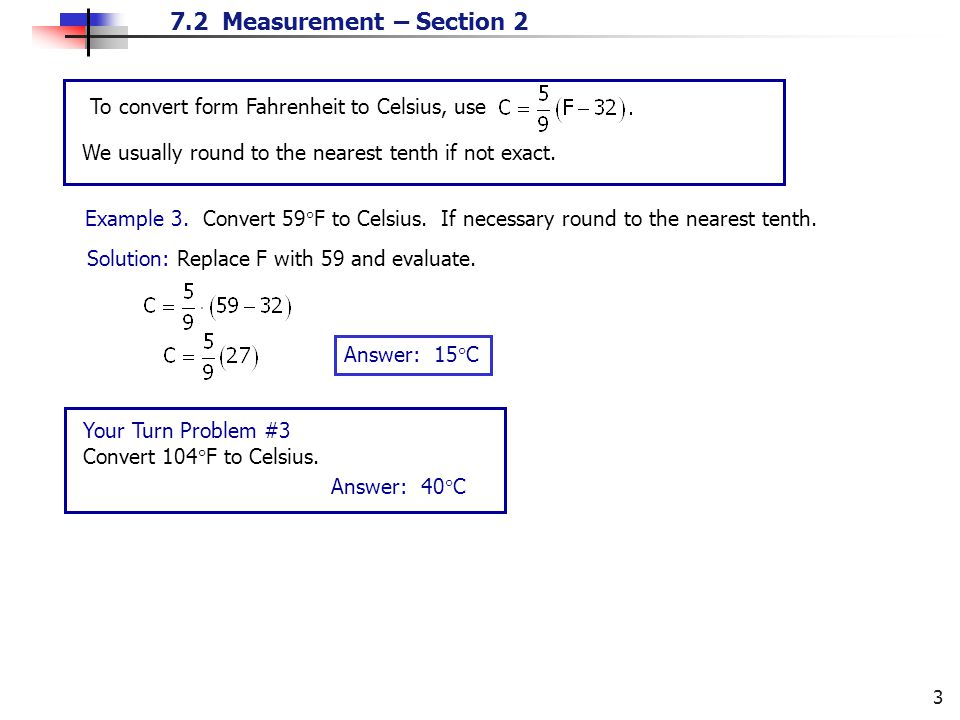
7.2 Measurement – Section 2 1 Objective A: To Convert between Celsius and Fahrenheit using the formulas There are two temperature scales: Fahrenheit, which. - ppt download

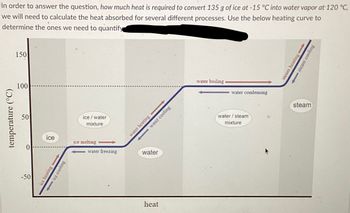
SOLVED: 1. How much heat is required to convert 135 g of ice at-15 'C into water vapor at 120 -C? Show all your work: Ciquid water 4.184 Jg %€ Csteam =

What amount of ice will remain when 52 g ice is added 100 g of water at 40^oC ?[Specific heat of water is 1 cal/g and latent heat of fusion of ice
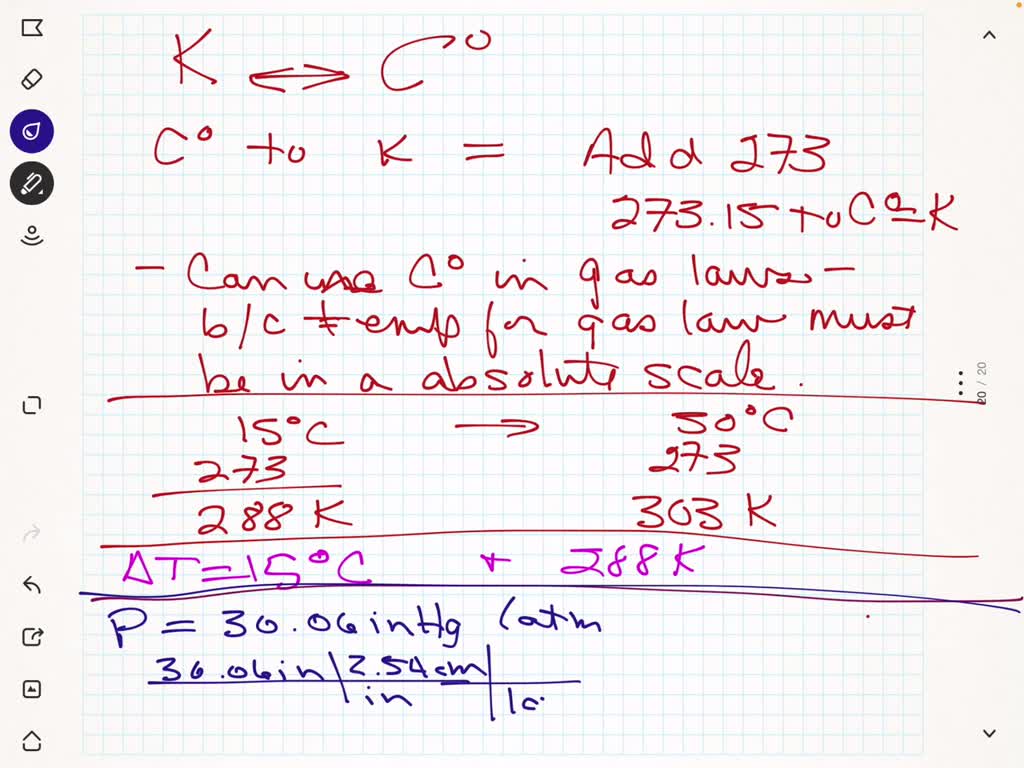
SOLVED: What is the conversion from Celsius to Kelvin and why is this conversion useful when applying gas law calculations? A room is heated from 15 °C to 30 °C. What are