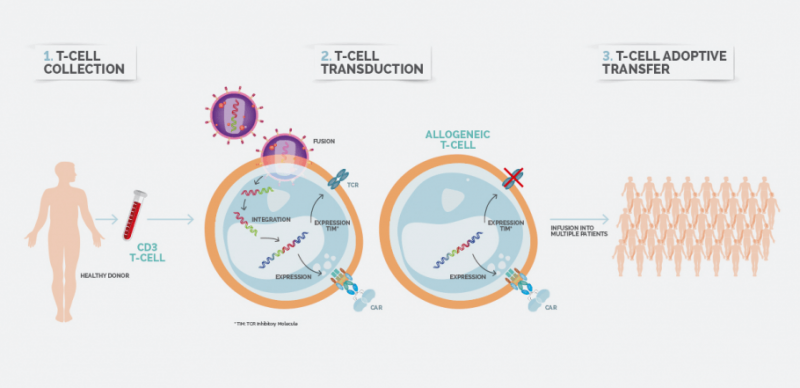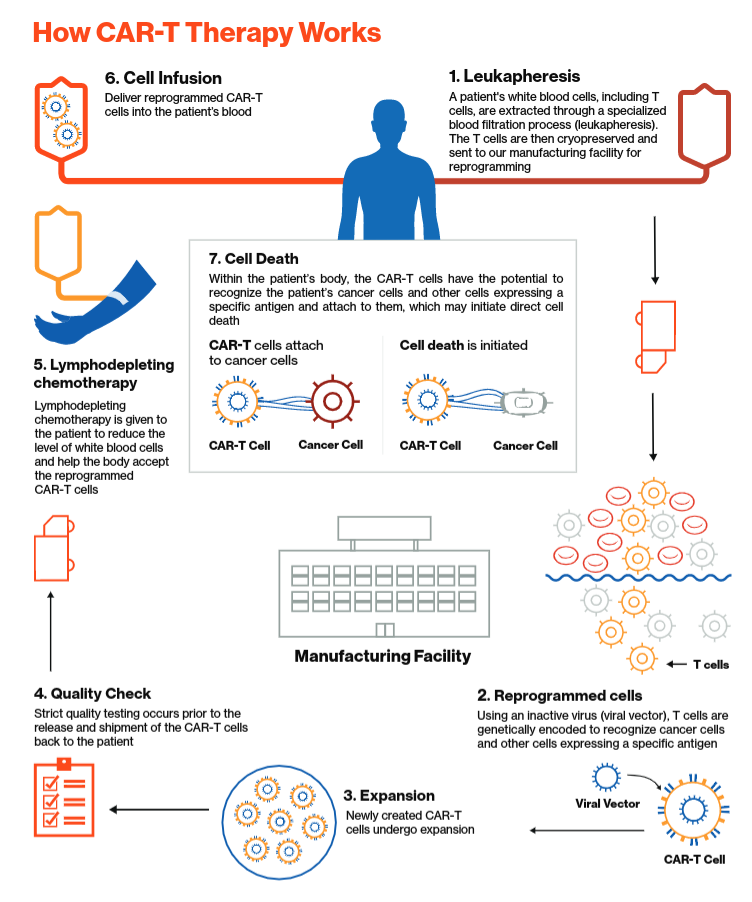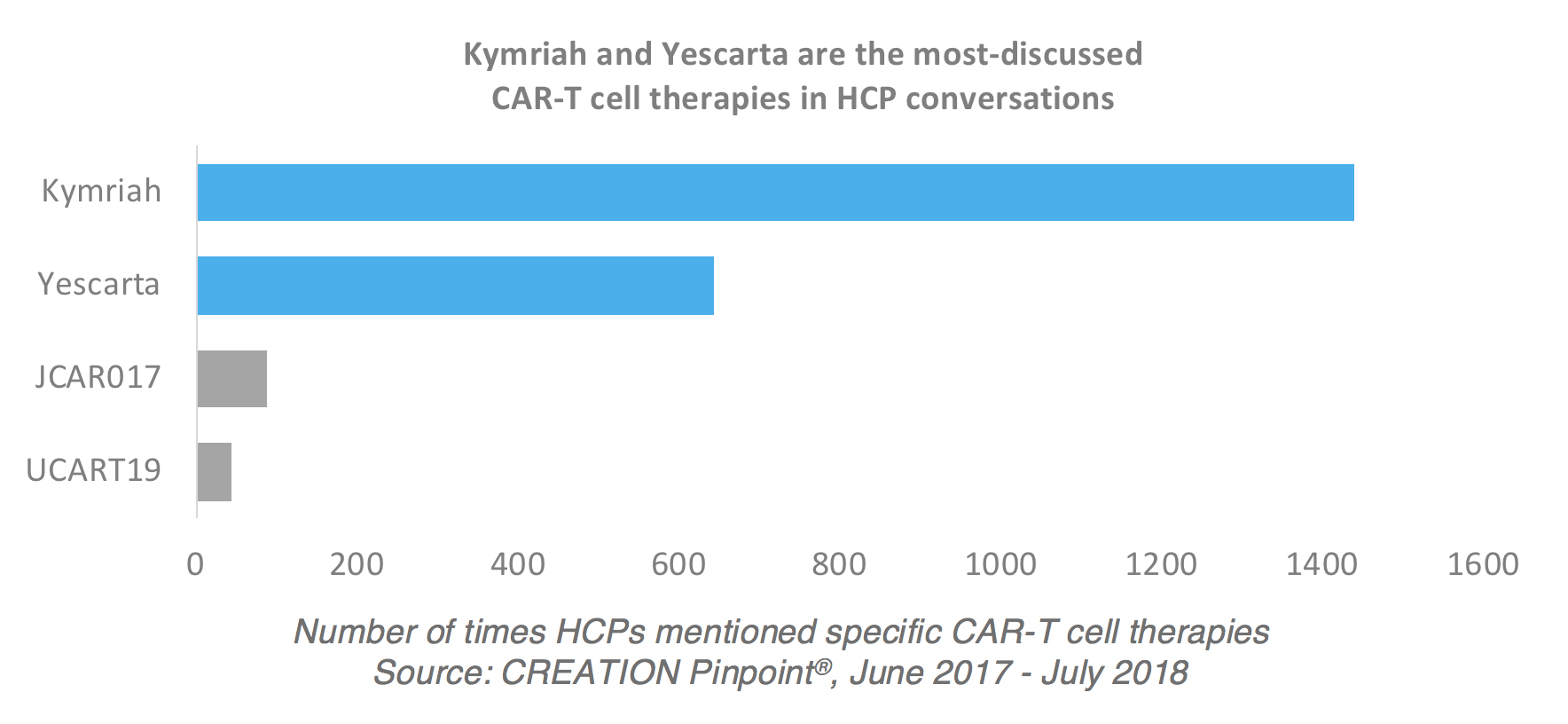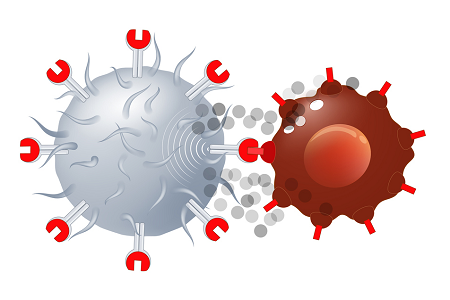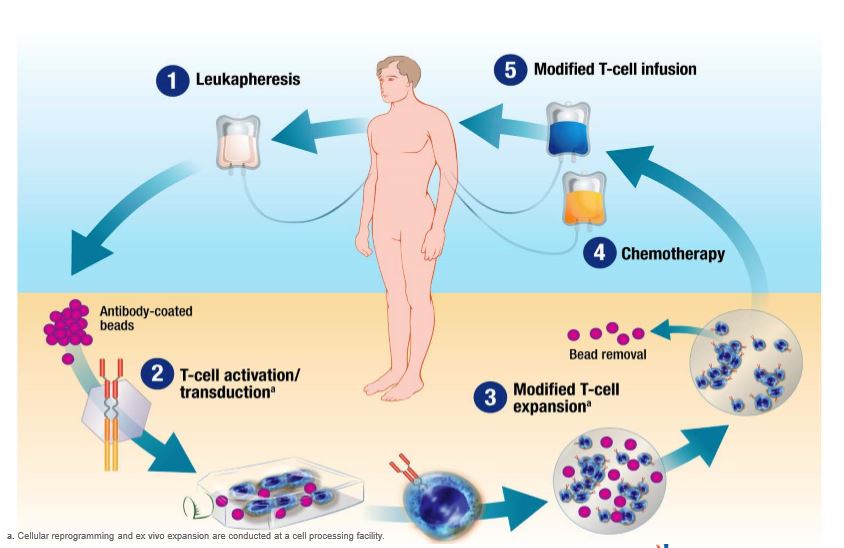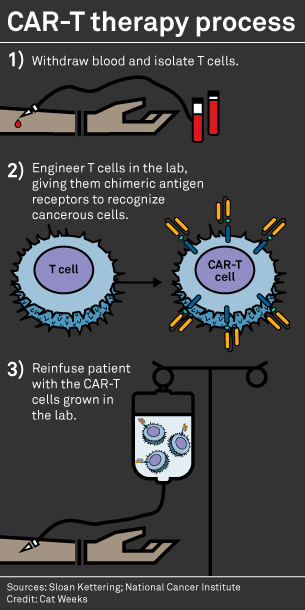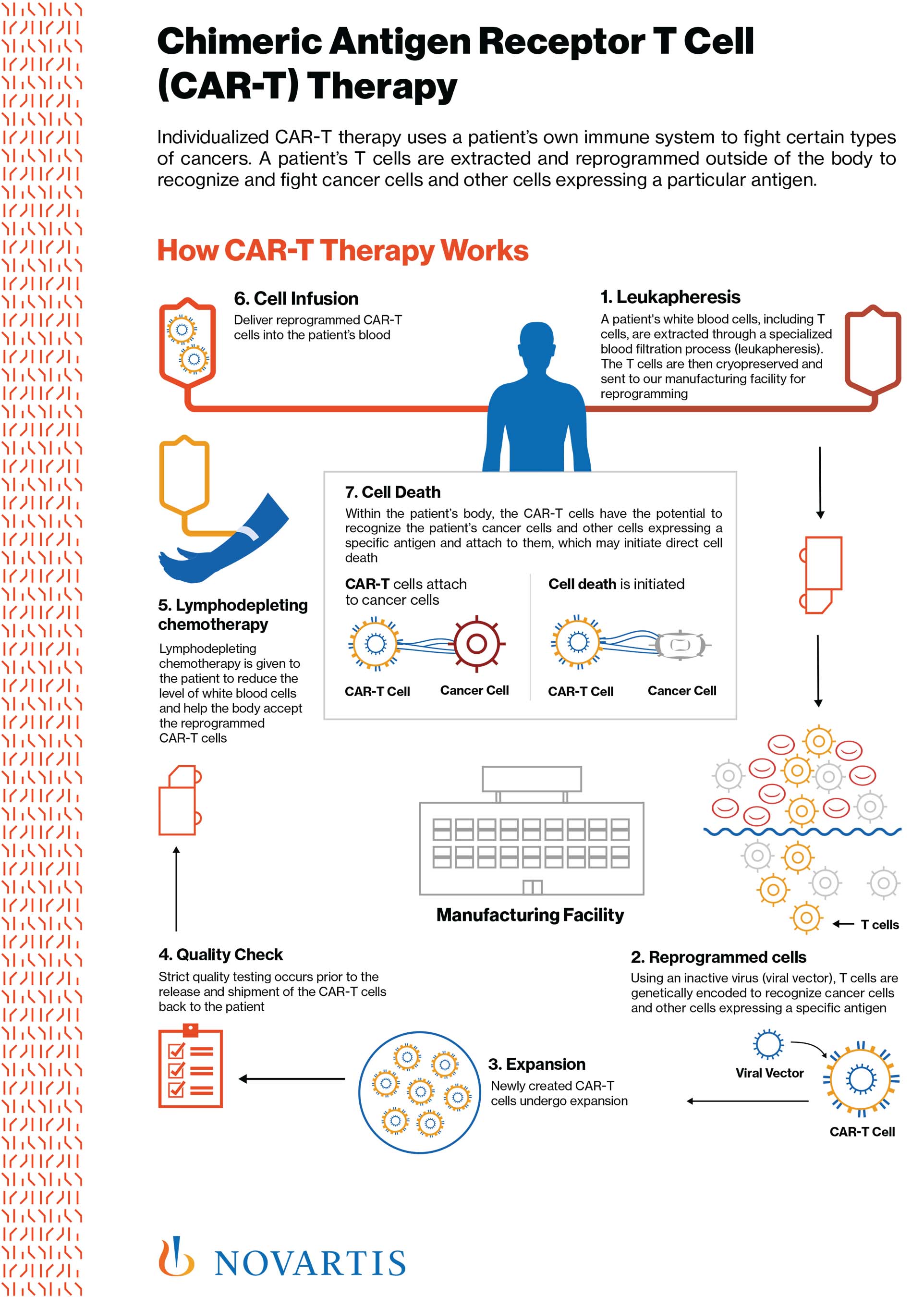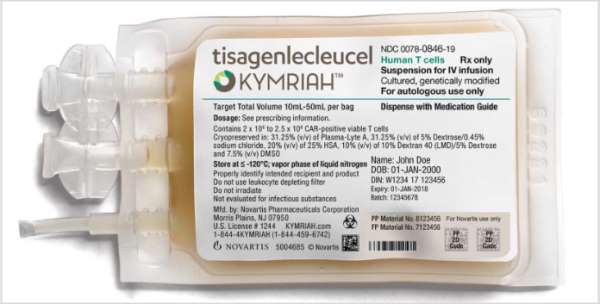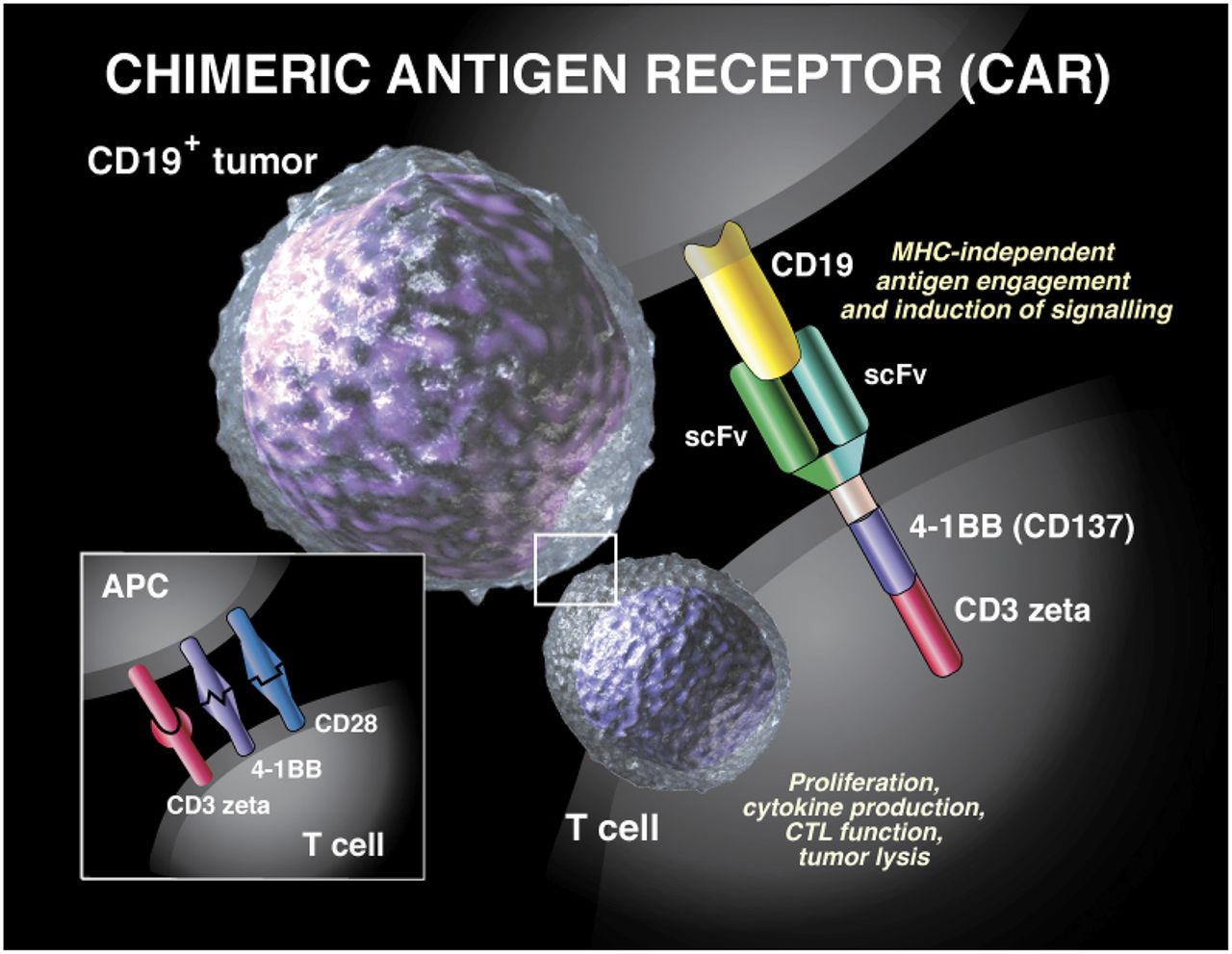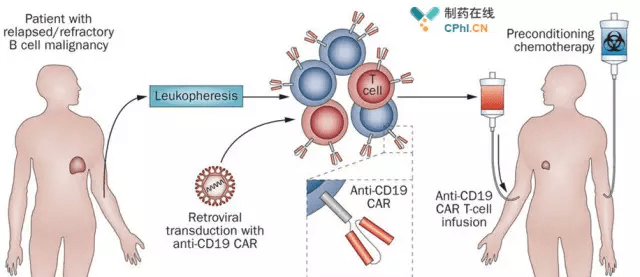
Historical moment! Milestone in immunotherapy: the world's first CAR-T cell product, CTL019, is approved for sale - Yongtai

Fraunhofer IZI and Novartis Continue Successful Collaboration with CAR-T Cell Therapy - Fraunhofer IZI
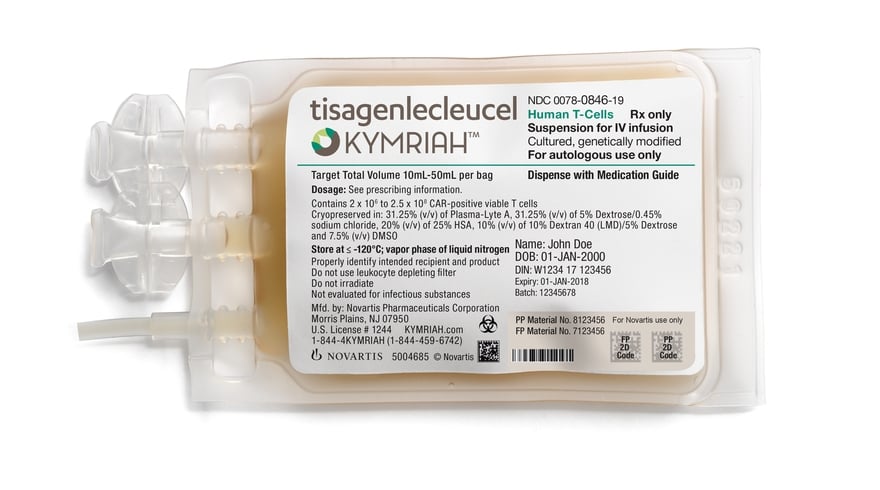
Novartis fills manufacturing gap for CAR-T therapy Kymriah with first Asian production facility | Fierce Pharma
Novartis receives first ever FDA approval for a CAR-T cell therapy, Kymriah(TM) (CTL019), for children and young adults with B-cell ALL that is refractory or has relapsed at least twice - IndiaMedToday
Anvisa aprova produto de terapia avançada para tratamento de câncer - ISMEP - Instituto Santa Marta de Ensino e Pesquisa