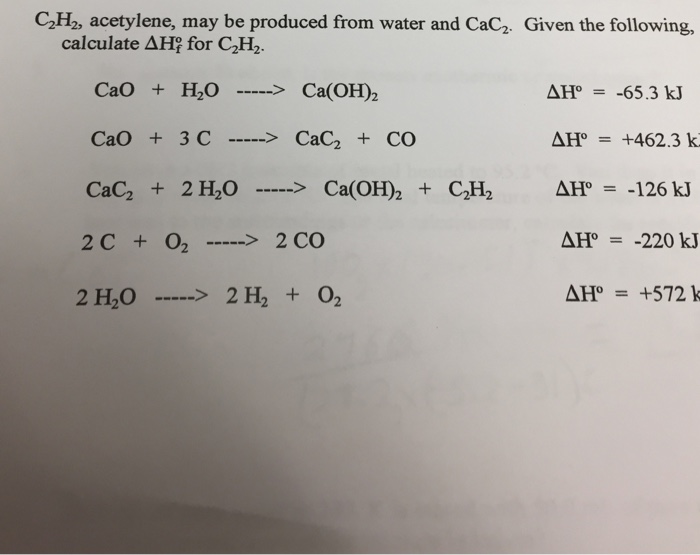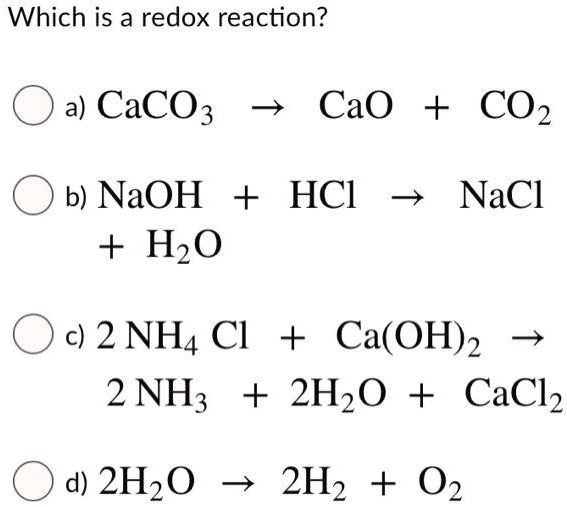
SOLVED: Which is a redox reaction? a) CaCO3 CaO + CO2 b) NaOH + HzO HCl NaCl c) 2 NH4 Cl + Ca(OH)2 2 NH; + 2Hz0 + CaClz d) 2Hz0 2H2 + 02

Insight into the role of CaO in coke-resistant over Ni-HMS catalysts for CO2 reforming of methane - ScienceDirect

Response FTIR spectra of CaO sample at (a) 700 °C, (b) 900 °C and (c)... | Download Scientific Diagram

Synthesis of novel eco-friendly CaO/C photocatalyst from coffee and eggshell wastes for dye degradation - ScienceDirect

CO2 Adsorption on CaO(001): Temperature-Programmed Desorption and Infrared Study | The Journal of Physical Chemistry C

Chemical Reactions Follow the matter… 1. Chemical Reactions Two chemicals have interacted in some way so that a new substance or substances are formed. - ppt download

i) CaO(s) + H2O (l) = Ca (OH)2(s) , Δ H180^oC = - 15.26 kcal (ii) H2O(l) = H2(g) + 12 O2 (g) , Δ H180^oC = 68.37 kcal (iii) Ca(s) +

End product of the following sequence is : CaO + C overset( "Heat")to (A) overset(H(2)O)to(c) underset(H(2)SO(4))overset(Hg^(2+))to(C)
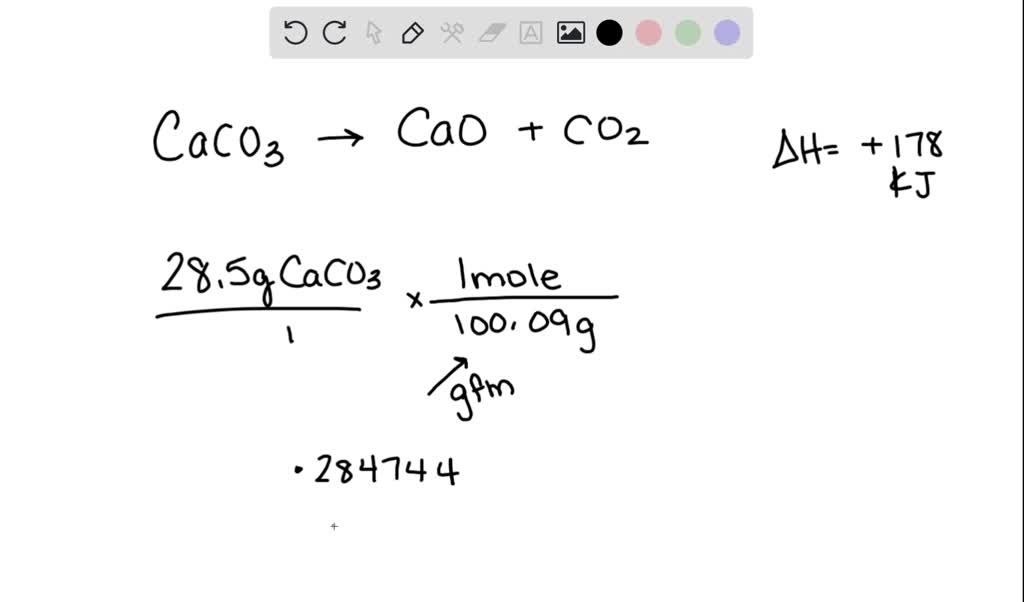
SOLVED: Consider the following thermochemical equation: CaCO3(s) CaO(s) + CO2 Ho = +178 kJ a) How many moles of CaCO3 are in 28.5g of CaCO3? moles b) How much heat must be
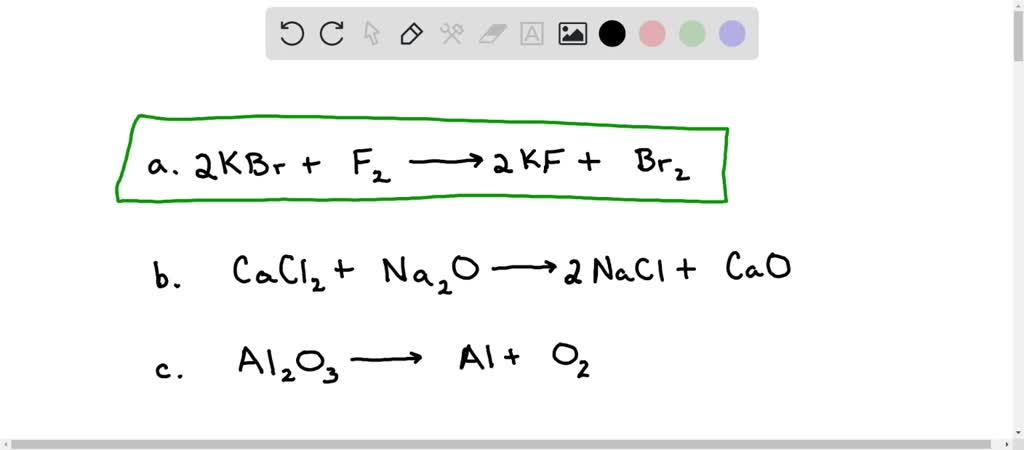
SOLVED: Balance the following reactions: a) KBr + F2 → KF + Br2 b) CaCl2 + Na2O → NaCl + CaO c) Al2O3 → Al + O2
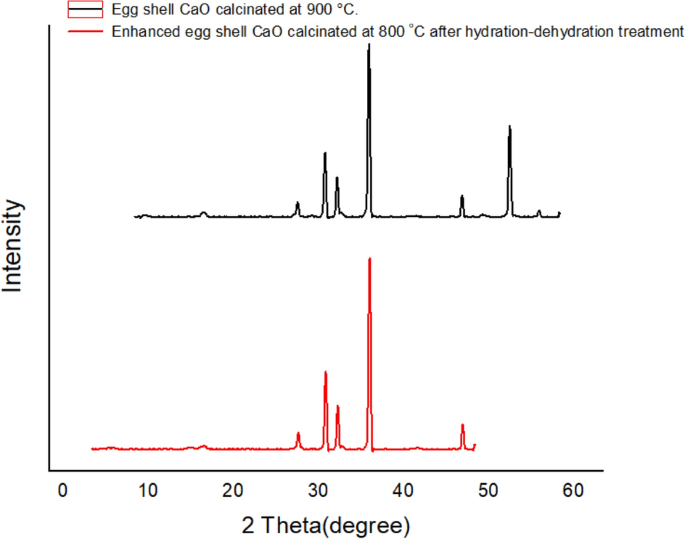
Improved biodiesel production from waste cooking oil with mixed methanol–ethanol using enhanced eggshell-derived CaO nano-catalyst | Scientific Reports
amino acid (1mole)gas evolvedsalt(0.1999 kg) amino acid having i.2 NH2 gr. ii.1 COOH gr iii.2 COOH gr how to deal with such problem? iv.3 COOH gr

Phase diagram of FexO-SiO2-CaO-MgO-"NiO"system, at PO2 = 10 −7 atm,... | Download Scientific Diagram

ПОМОГИТЕ!!! CaO---(C, 2000 градусов)---> A --(H2O)-->Б --(C, 450 градусов)--> B - Школьные Знания.com