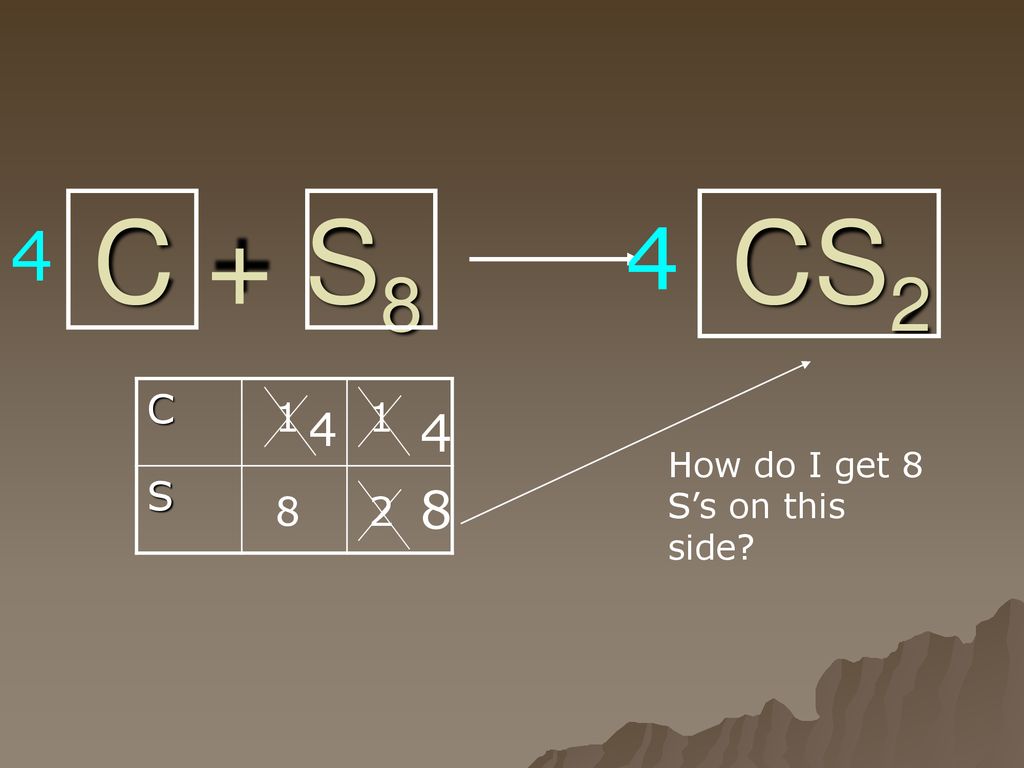
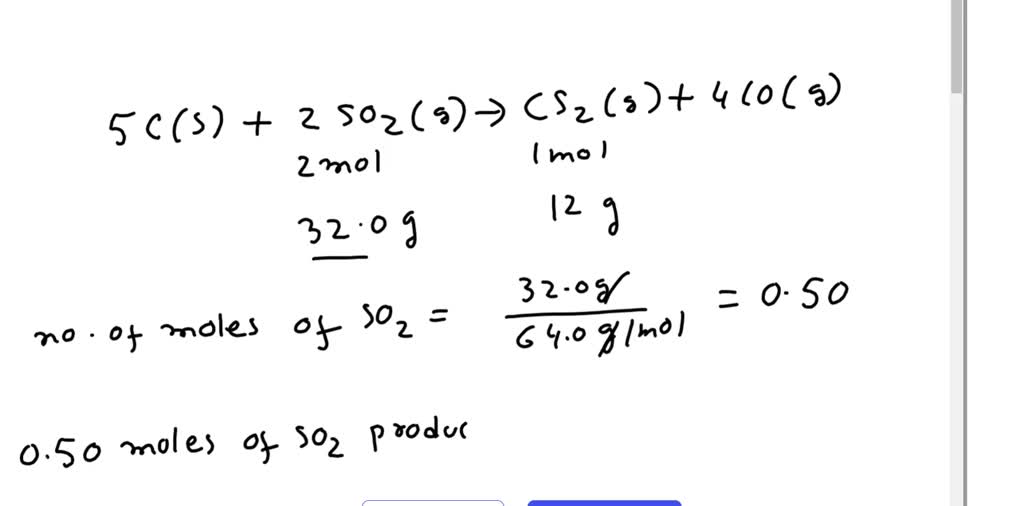C+S8=CS2 balance the chemical equation @mydocumentary838. c+s8=cs2 balance the chemical equation. - YouTube
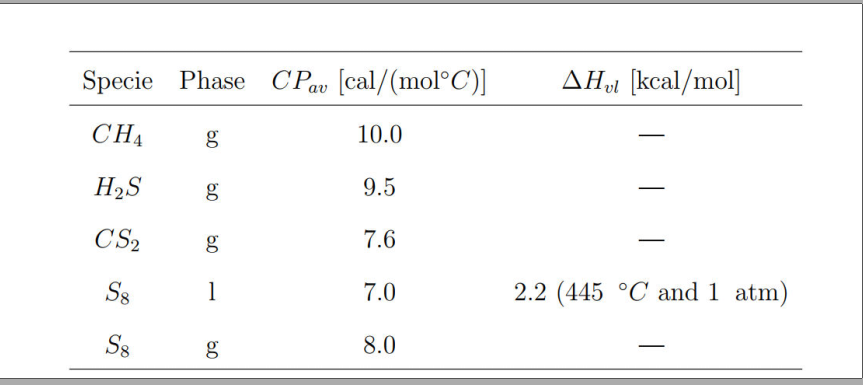
![SOLVED: For the [Referencos] following reactions determine the value of € (in mol) 4C + S8 CS2 1.9 mol C yields € mol CSz mol CSz + 3 02 COz + 2 SOLVED: For the [Referencos] following reactions determine the value of € (in mol) 4C + S8 CS2 1.9 mol C yields € mol CSz mol CSz + 3 02 COz + 2](https://cdn.numerade.com/ask_images/011d73f2dcd2412388390021a974e15a.jpg)
SOLVED: For the [Referencos] following reactions determine the value of € (in mol) 4C + S8 CS2 1.9 mol C yields € mol CSz mol CSz + 3 02 COz + 2

C+S8=CS2 balance the chemical equation @mydocumentary838. c+s8=cs2 balance the chemical equation. - YouTube

Photocatalytic splitting of CS2 to S8 and a carbon-sulfur polymer catalyzed by a bimetallic ruthenium(II) compound with a tertiary amine binding site: toward photocatalytic splitting of CO2? | Semantic Scholar