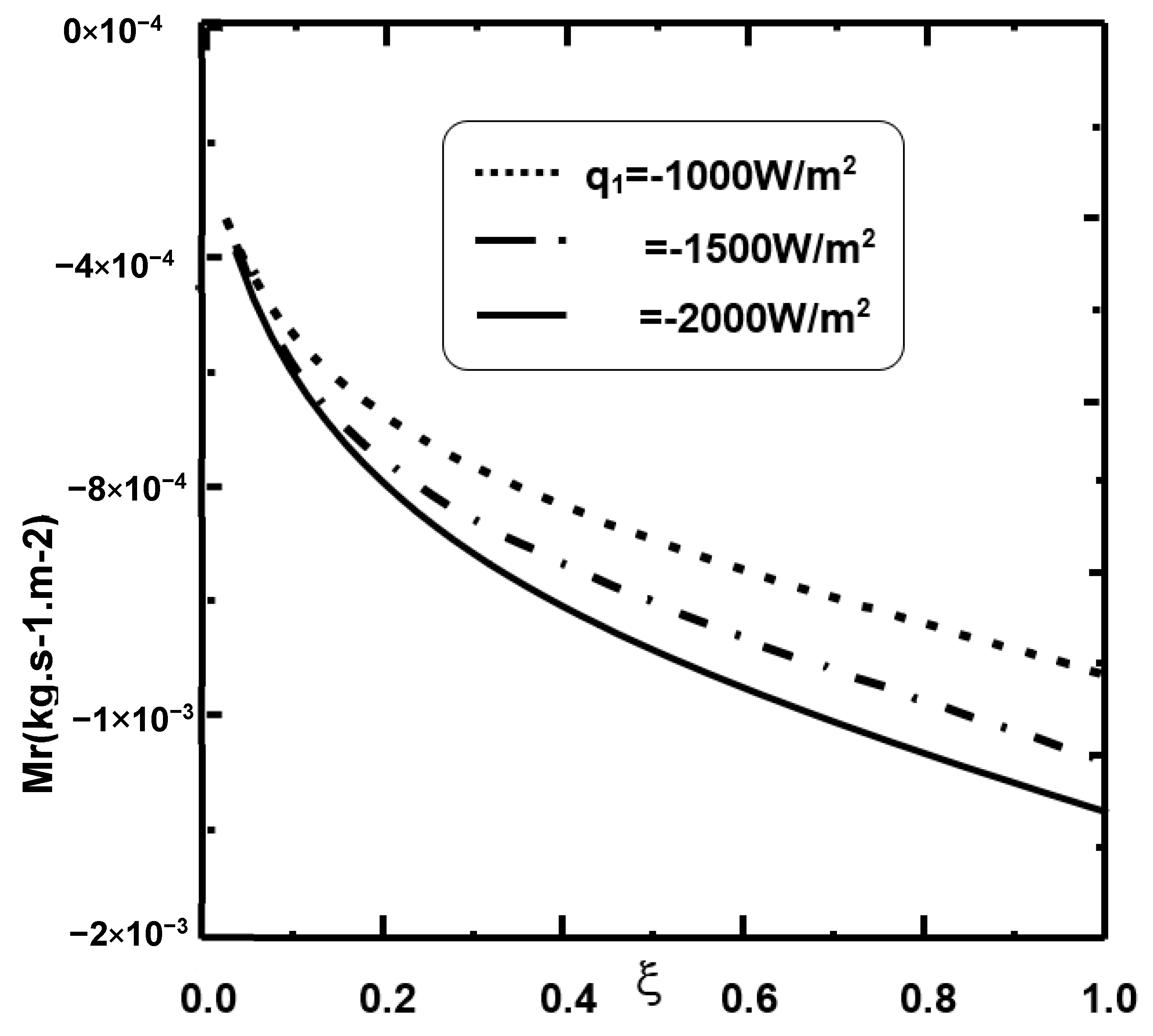
Applied Sciences | Free Full-Text | Liquid Nanofilms’ Condensation Inside a Heat Exchanger by Mixed Convection

Densities, Viscosities, and Electrical Conductivities of Pure Anhydrous Reline and Its Mixtures with Water in the Temperature Range (293.15 to 338.15) K | Journal of Chemical & Engineering Data

A) Dilatational modulus (E) for C 12 SO 4 − at 283.15 K and 293.15 K... | Download Scientific Diagram

Thermodynamics of mixtures containing amines. XIV. CpmE of benzylamine with heptane at 293.15 K or with methanol, 1-propanol or 1-pentanol at 293.15–308.15 K - ScienceDirect

Density and Excess Molar Volumes of 1-Butanol + Methanol + Electrolyte Systems in the Temperature Range 293.15–308.15 K | Journal of Chemical & Engineering Data

Thermodynamics and Kinetic Studies for the Adsorption Process of Methyl Orange by Magnetic Activated Carbons - Ana Karen Cordova Estrada, Felipe Cordova Lozano, René Alejandro Lara Díaz, 2021

OpenStax College Physics Solution, Chapter 15, Problem 50 (Problems & Exercises) | OpenStax College Physics Answers

OpenStax College Physics for AP® Courses Solution, Chapter 13, Problem 4 (Test Prep for AP® Courses) | OpenStax College Physics Answers
Experimental Study and Mathematical Modeling of a Nanofiltration Membrane System for the Recovery of Polyphenols from Wine Lees






:max_bytes(150000):strip_icc()/76celcius-58ed113e5f9b58f1194321cb.jpg)

